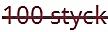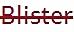Under knappen Förpackningar i Fass har vi samlat praktisk information om förpackningar och priser.
Här får du veta bland annat vilka olika förpackningar läkemedlet levereras i, om de är receptbelagda och vad läkemedlet kostar.
Observera att läkemedel till djur inte omfattas av läkemedelsförmånen, och att fri prissättning gäller, därav kan du ej hitta prisuppgifter på Fass för dessa.
I tabellen får du bland annat reda på:
-
Läkemedelsförpackningarnas varunummer, storlek, typ och om de för tillfället marknadsförs eller inte.
-
Om läkemedlet är receptfritt eller receptbelagt, och om någon utöver läkare får förskriva läkemedlet.
-
Om läkemedlet ingår i läkemedelsförmånen.
-
Om det finns förpackningar som parallelldistribuerade av andra företag visas information om detta.
Några saker som kan vara bra att veta:
-
Priser inom läkemedelsförmånen.
Läkemedelsförpackningar som ingår i läkemedelsförmånen har ett förmånspris när du köper dem med recept. Priset fastställs av Tandvårds- och läkemedelsförmånsverket (TLV). Det aktuella priset visas alltid på Fass.
-
Läkemedel utanför läkemedelsförmånen.
Läkemedel utanför läkemedelsförmånen har fri prissättning. Priserna varierar mellan olika apotek och kan därmed inte anges på Fass. Det gäller även receptfria läkemedel och läkemedel till djur.
Presentation av tabellkolumnerna
En kommentar skrivs bara ut en gång fast den kan gälla även för senare exempel.
a) Receptfri förpackning med förmånspris på recept utan extra förskrivarkategorier
|
Storlek
(1)
|
Förpacknings-typ
(2)
|
Förmåns-pris med recept(3)
|
Recept-belagd (4)
|
Övriga för-skrivare (5)
|
Varu-nummer (6)
|
|
100 st
|
Blister
|
199:50
|
Nej
|
-
|
012345
|
-
Kolumn 1: Anger förpackningens storlek med avseende på kvantitet eller volym.
-
Kolumn 2: Anger förpackningens typ. Denna kan exempelvis utgöras av en burk, tryckförpackning (blister), tub etc.
-
Kolumn 3: Berättar att läkemedelsförpackningen ingår i läkemedelsförmånen på recept och har fått ett nationellt försäljningspris av Tandvårds- och Läkemedelsförmånsverket.
-
Kolumn 4: Visar att läkemedels-förpackningen inte är receptbelagd och kan köpas i apotekens egenvårdsavdelningar eller i vissa fall i dagligvaruhandeln (fri prissättning).
-
Kolumn 5: Inga förskrivargrupper utöver läkare får förskriva recept på läkemedelsförpackningen.
-
Kolumn 6: Anger förpackningens varunummer
B) Receptbelagd förpackning med förmånspris som har extra förskrivarkategorier
|
Storlek
(1)
|
Förpacknings-typ
(2)
|
Förmåns-pris med recept(3)
|
Recept-belagd (4)
|
Övriga för-skrivare (5)
|
Varu-nummer (6)
|
|
100 st
|
Blister
|
199:50
|
Ja
|
Sjuksköterska Tandhygienist Tandläkare
|
012345
|
-
Kolumn 4: Förpackningen är receptbelagd.
-
Kolumn 5: Får förskrivas även av sjuksköterskor med förskrivningsrätt, tandhygienister och tandläkare.
C) Receptbelagd förpackning med villkorat förmånspris.
|
Storlek
(1)
|
Förpacknings-typ
(2)
|
Förmåns-pris med recept
(Med villkor)(3)
|
Recept-belagd (4)
|
Övriga för-skrivare (5)
|
Varu-nummer (6)
|
|
100 st
|
Blister
|
199:50
|
Ja
|
-
|
012345
|
D) Receptfri förpackning utan förmånspris på recept som inte tillhandahålls.
|
Storlek
(1)
|
Förpacknings-typ
(2)
|
Förmåns-pris med recept
(3)
|
Recept-belagd (4)
|
Övriga för-skrivare (5)
|
Varu-nummer (6)
|
|
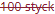
|
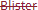
|
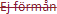
|
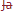
|
-
|

|
E) Receptbelagd förpackning utan förmånspris.
|
Storlek
(1)
|
Förpacknings-typ
(2)
|
Förmåns-pris med recept
(3)
|
Recept-belagd (4)
|
Övriga för-skrivare (5)
|
Varu-nummer (6)
|
|
100 st
|
Blister
|
Ej förmån
|
Ja
|
-
|
012345
|
F) Receptfri veterinär förpackning.
|
Storlek
(1)
|
Förpacknings-typ
(2)
|
Recept-belagd (3)
|
Varu-nummer (4)
|
|
100 st
|
Blister
|
Nej
|
123456
|
• Kolumn 1: Anger förpackningens storlek med avseende på kvantitet eller volym.
• Kolumn 2: Anger förpackningens typ. Denna kan exempelvis utgöras av en burk, tryckförpackning (blister), tub etc.
• Kolumn 3: Visar att läkemedels-förpackningen inte är receptbelagd och får köpas i egenvården. (Apoteken bestämmer själva försäljningspriset).
• Kolumn 4: Anger förpackningens varunummer.
G) Receptbelagd veterinär förpackning.
|
Storlek
(1)
|
Förpacknings-typ
(2)
|
Recept-belagd (3)
|
Varu-nummer (4)
|
|
100 st
|
Blister
|
Ja
|
123456
|
• Kolumn 3: Visar att läkemedelsförpackningen är receptbelagd. (Apoteken bestämmer själva försäljningspriset).
G) Receptbelagd veterinär förpackning som inte tillhandahålls för närvarande.
|
Storlek
(1)
|
Förpacknings-typ
(2)
|
Recept-belagd (3)
|
Varu-nummer (4)
|
|
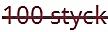
|
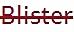
|
Ja
|
123456
|
• Kolumn 1 och 2: Den röda textöverstrykningen i kolumnerna "Storlek" och "Förpackningstyp" anger att förpackningen inte marknadsförs/tillhandahålls för närvarande.
Texten reviderades senast: 2024-10-10
Publiceringsdatum: 2024-10-10
|